
A new study shows that lowering MSH3, a key DNA repair protein, with antisense oligonucleotide (ASO) therapy can stop CAG repeat expansions in HD patient-derived brain cells. This could be a promising way to slow symptom onset and progression.

Disappointing news from Roche and Ionis; the phase III Tominersen huntingtin-lowering trial has been halted early

Scientists screen the ENTIRE genome to find new potential therapeutic targets for HD. This ambitious study provides a wealth of data for HD researchers
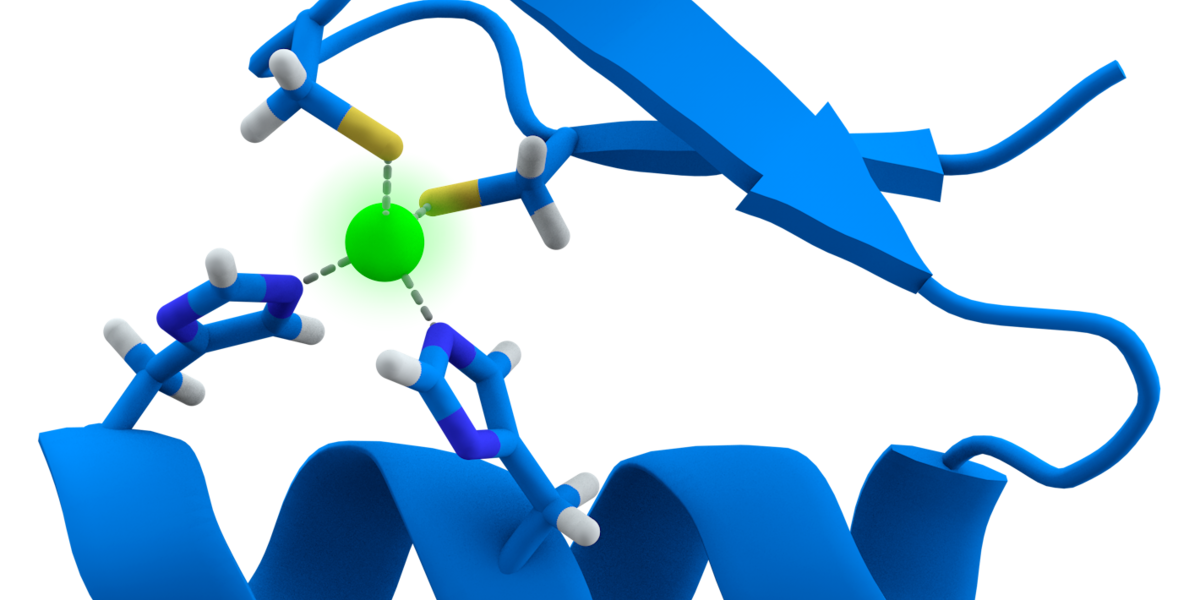
Exciting new Huntingtin Lowering work from @SangamoTx and @CHDIfoundation using "Zinc Fingers" to shut down expression of the mutant Huntingtin gene. More details on this exciting new technique here.
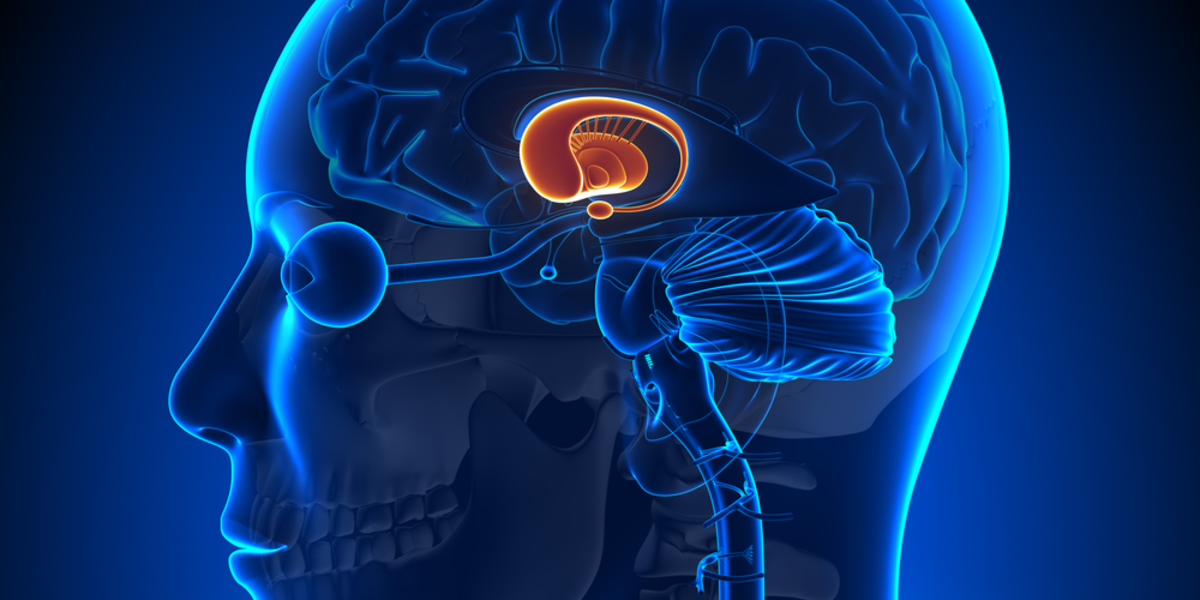
UniQure announces key details of its planned trial to assess the safety and ability of AMT-130 gene therapy to lower the problematic huntingtin protein using a ‘single-shot’ virus delivery system.

Hot off the presses - New publication gives more details about the results of Ionis and Roche's safety study with a Huntingtin-lowering ASO
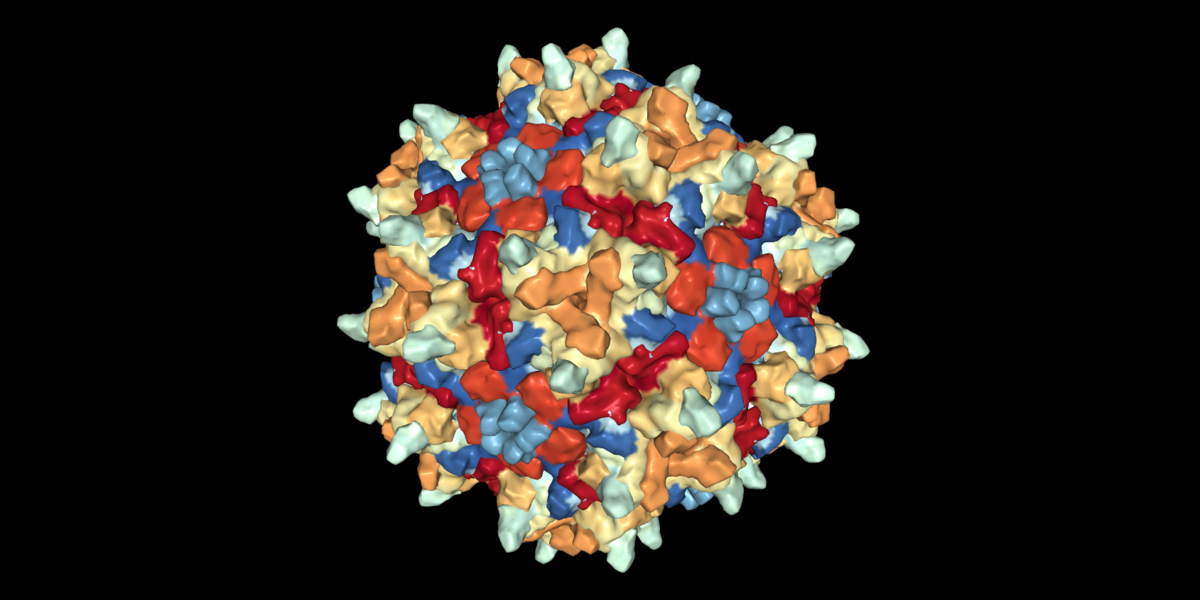
FDA grants “Investigational New Drug” status to Huntingtin-lowering gene-therapy agent AMT-130, clearing path to human trials in Huntington’s Disease patients

This fall sees exciting announcements from a number of companies focused on novel Huntingtin Lowering technologies, including Wave, PTC and Voyager

HDSA's frequently asked questions factsheet about the huntingtin-lowering trial ASO program was so good we stole it (with permission)