
Cell replacement therapy is making massive strides for other diseases. But what about Huntington’s disease? Can we use technological advancements in this area to replace lost brain cells with the hope of repairing and reversing disease?

Apathy in Huntington’s disease isn’t just a lack of motivation—it’s a shift in how the brain weighs decisions. A new study sheds light on how targeted treatments could help restore motivation and improve quality of life.

As we begin 2025, we look back on all of the Huntington’s disease research news and progress the field has made in the last 12 months.

We’re proud to announce AJ Keefe as our last HDBuzz Prize winner of 2024! Our cells are an intricate choreography between various molecular dancers. Knowing who moves well with whom, and how that changes with disease, could help advance drugs for HD.

Vico Therapeutics have shared interim data about their drug, VO659, which targets the CAG expansion that causes several genetic diseases, including #Huntingtonsdisease and #spinocerebellarataxia

In an update shared today, uniQure announced alignment with the US drug regulator on key criteria for accelerated approval of drugs for Huntington’s disease.

A new Phase 1 clinical trial for the huntingtin-lowering drug ALN-HTT02 was initiated this week with the first dose given. Read on to learn details about the trial and how it compares to other ongoing huntingtin-lowering trials in the clinic.

At this time of giving, we want to give thanks to you, the Huntington’s disease (HD) community, for all you have done for HD research, bringing us closer to medicines which will one day slow or halt HD.

Advanced Care Planning is a tool that can allow people living with Huntington’s disease to compassionately engage with loved ones about their future care, taking thoughtful steps at their own pace while they’re able, to ensure their wishes are met.
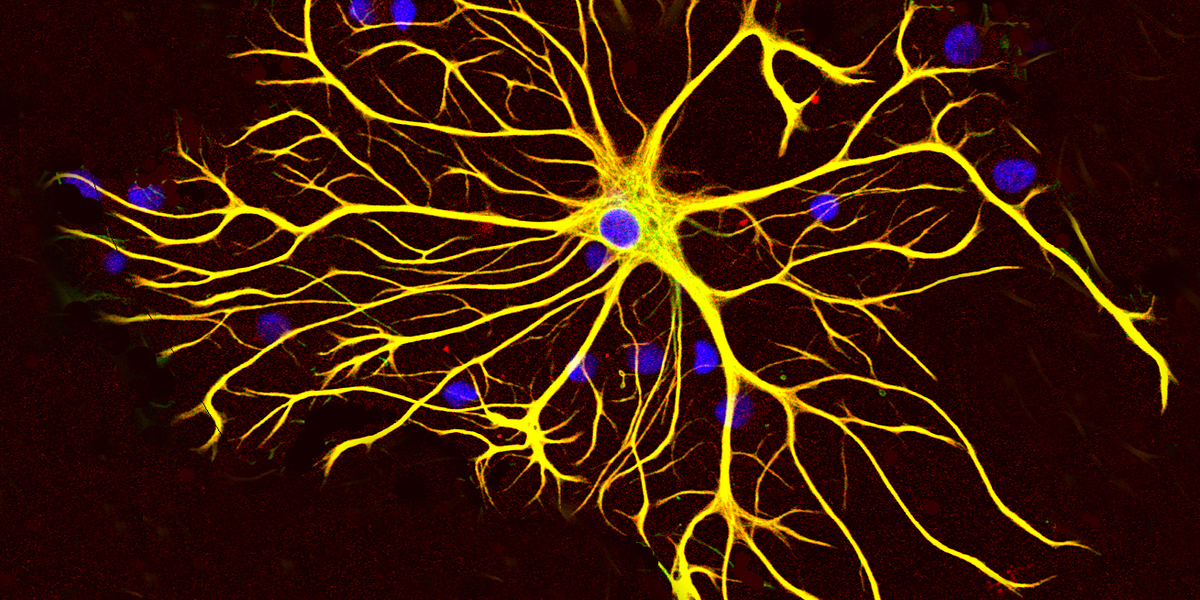
We're proud to announce Jenny Lange as a 2024 HDBuzz Prize winner! Our brains are made up of many types of cells that play different roles in the changes caused by HD. This study dives into a star-shaped cell called astrocytes and their role in HD.

The HDSA recently coordinated a meeting between Huntington’s disease families and the US regulatory agency that approves medicines, to amplify the community’s voice and help move us toward treatments.