The brain in Huntington's disease: greater than the sum of its parts?
An important new study asks the question: what parts of the brain might need most help in HD?

The symptoms of HD are caused by damage to the brain, but not all parts of the brain are affected equally. This raises an important question – if we had a treatment that could help only a small part of the brain, which part would we pick? A new mouse study from William Yang, at UCLA, attempts to answer this question.
What brain region causes HD?
The major symptoms of Huntington’s disease are familiar to most HD family members: decline in thinking ability, increased emotional problems and movement disturbances. Scientists believe that these problems all have their origin in the malfunctioning and loss of cells in the brain.
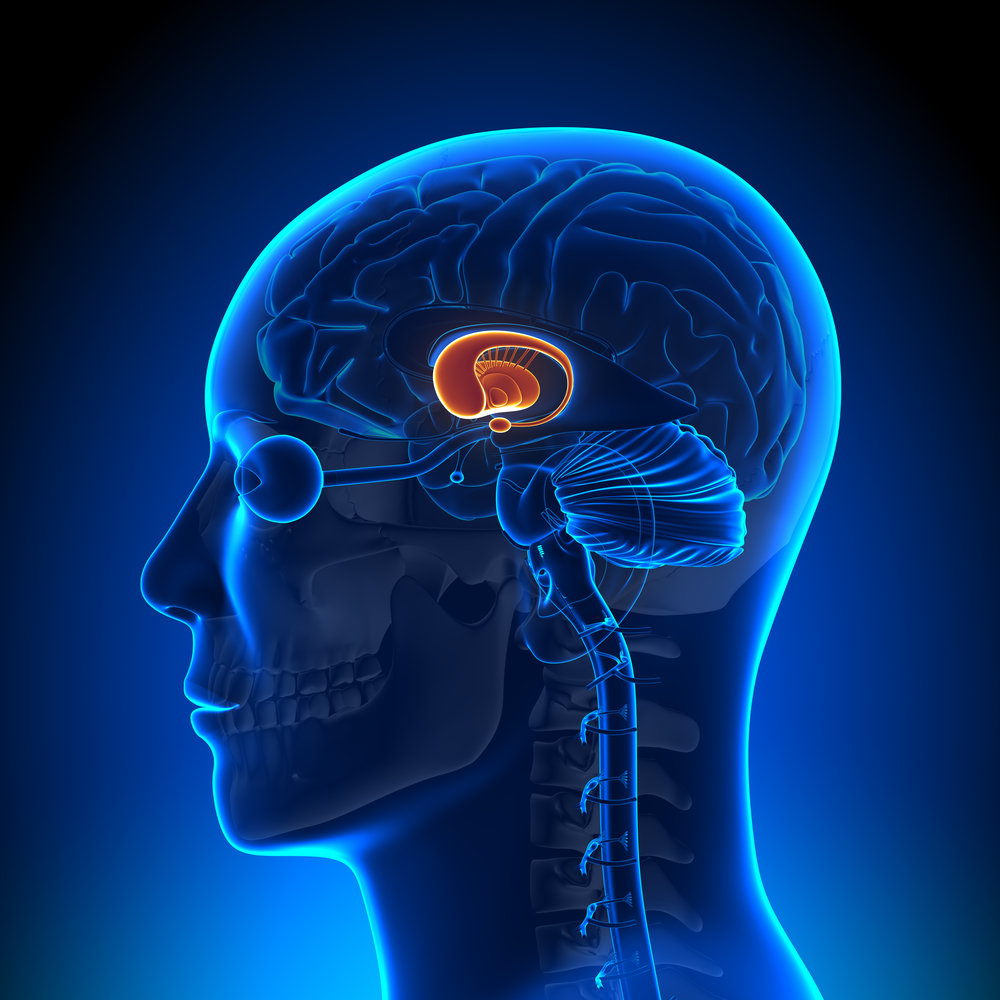
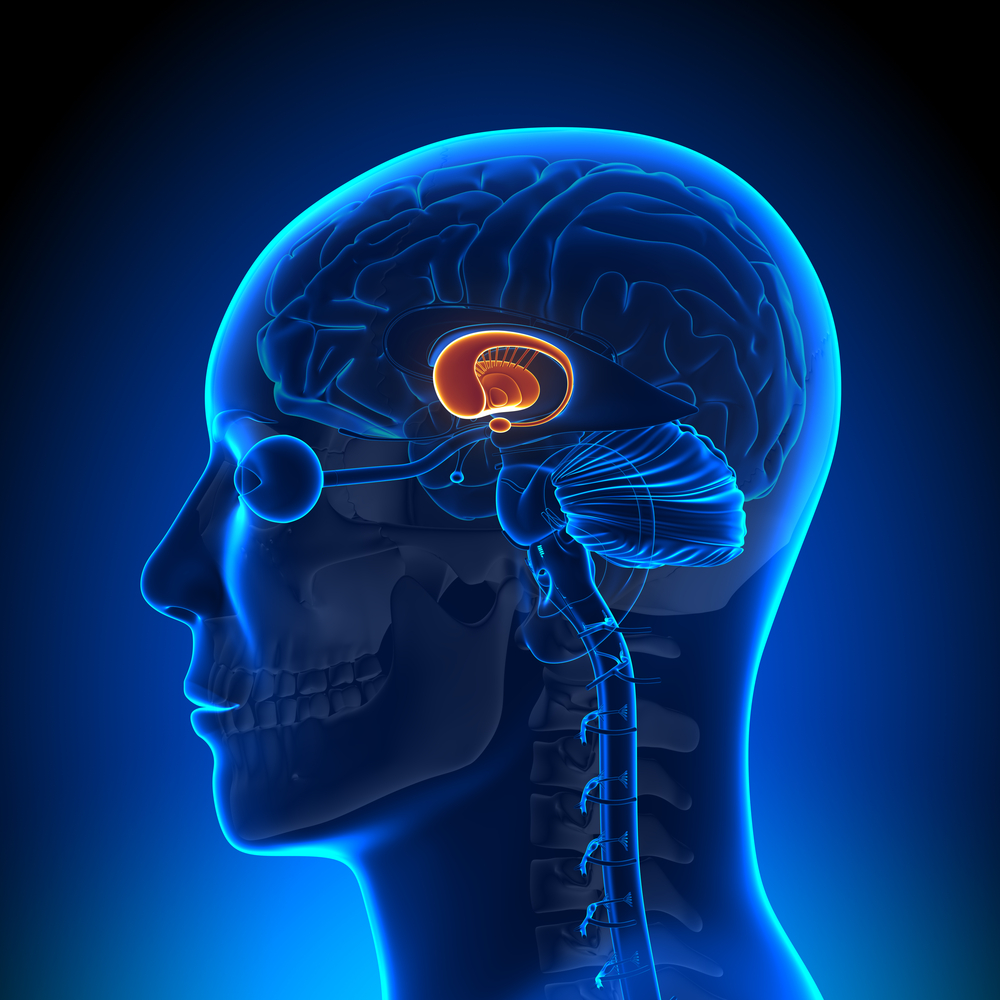
But not just anywhere in the brain – the pattern of cell loss in HD is very specific. Given the brains of people who died from a range of brain diseases, a talented doctor could tell you which died from HD, compared to patients who died from Alzheimer’s or Parkinson’s disease. They can do this because, in each case, the disease is associated with some parts of the brain being affected more prominently than others.
The most vulnerable part of the brain in HD is called the striatum. The striatum is a relatively small structure, deep underneath the wrinkly outside part of the brain, which is called the cortex.
During the course of HD, cells in both the striatum and cortex shrink, dysfunction and eventually die. Study after study, investigating many hundreds of volunteers, has found that the striatum is the first location in the brain that shrinks in people carrying the HD mutation.
Brain connectivity
In many ways, the brain is unique compared to other organs in your body. One way the brain is unique is that the cells that help us think, called neurons, are heavily connected to each other. On average, each of the 100 billion neurons in the brain is connected to thousands of other neurons – giving each human brain as many as 100 trillion connections!
The connections in the brain are not random: specific parts of the brain know that their job is to talk to one another. You want to make sure that the optic nerve coming out of the back of your eye is properly attached to the part of your cortex that handles vision, for example.
In this way, the striatum and cortex are very tightly connected – in fact, the cortex sends maybe billions of connections down to the striatum. Interestingly, the connections don’t go in both directions – the striatum sends its own connections somewhere else in the brain.
Being connected to one another isn’t just how neurons stay in communication with each other, it’s actually how they stay alive. For many years scientists have known that neurons stripped of their connections with other neurons will actually die!
“Is treating the striatum enough? The answer, based on the best data we have so far, is “probably not”. ”
This raises interesting questions in HD. Given that both the cortex and the striatum seem to shrink in HD, which tissue is responsible for what particular symptoms of HD? Does loss of the striatum (the deep part of the circuit) lead to loss of the cortex, or vice versa?
Mouse tricks
These kinds of questions can’t be answered in human beings, but we can use mice to try and get to the bottom of what’s happening in the Huntington’s disease brain. A team of scientists led by William Yang, at UCLA, used a special kind of HD mice to try and understand these issues.
The mice used by William have been genetically manipulated to have a mutant HD gene (though normal mice never get HD). Yang’s team gave these mice a special genetic quirk – their mutant HD gene can be turned off in specific parts of the brain, chosen by the scientists breeding the mice.
This gives us the ability to compare three different HD mice: ‘regular’ HD mice, with the abnormal active throughout the brain; HD mice with no mutant HD gene in their striatum; and HD mice with no mutant HD gene in their cortex. By comparing these groups, we can try to understand which brain region is most important for producing the symptoms of HD, at least in this mouse model.
Findings
Yang’s group turned to a number of tests used in labs to measure mouse behavior that are already known to be changed in HD mice. In these tests the HD mice performed worse than normal mice, as expected.
Based on the fact that the striatum is the most affected part of the brain in HD patients, we might expect that getting rid of the mutant HD gene here would be of the most benefit to the mice. Getting rid of the mutant HD gene in the striatum did lead to some improvement in the behavior of HD mice, though the numbers were a little low to be certain.

Surprisingly, mice lacking the mutant HD gene in the cortex (which is affected later in people) seemed to do better than HD mice lacking the HD gene in the striatum – suggesting the cortex is really important in HD.
A similar trend was seen when the team looked at changes in the physical structure of the brain, which again revealed that HD mice with no mutant HD gene in their cortex looked better than HD mice without a mutant HD gene in the striatum.
Finally, Yang’s team noted that to completely prevent the symptoms of HD, it was necessary to turn off the gene in the cortex and striatum.
Implications
This study suggests that the mutant Huntington’s disease protein causes problems in both the cortex and the striatum – and that both regions are important.
Any therapies that target only the striatum might have limited effectiveness, given the remaining problems elsewhere in the brain. This question is not just academic – certain proposed therapies for HD, including some forms of gene therapy and stem-cell replacement, will likely target only the striatum.
This work relies on genetic tricks that aren’t possible in humans, so it doesn’t directly point to a therapy for people. But it does use these tricks to uncover an answer to a question that HD scientists have argued about for a long time: “is treating the striatum enough?” The answer, based on the best data we have so far, is “probably not”.
So why are we excited about this new work? Because we’d rather learn these lessons in mice, and use them to design the best possible studies in humans with HD. This work is an important piece of the puzzle, and hopefully it’ll help us design the best possible clinical trials in the future.
Learn more
For more information about our disclosure policy see our FAQ…


