
Hunting for balance: how the huntingtin protein compensates in HD
Researchers look at the cause and effect of various forms of the HTT protein. They find both expanded and unexpanded HTT contribute to brain cell communication and the brain has an amazing capacity to compensate for changes related to disease


A group led by Dr. Sandrine Humbert from the French National Institute for Health and Medical Research published new work in the prestigious journal Science. Dr. Humbert’s team did some really cool science in mice to look at how both the expanded and unexpanded copies of huntingtin (HTT) affect mouse “symptoms” of Huntington’s disease (HD). Let’s break down the experiments they did and what their findings suggest.
Different forms of HTT contribute to HD
The genetic cause of HD is an extra stretch of DNA in a gene called Huntingtin (HTT) that leads to an expanded form of the HTT protein. The vast majority of people with HD inherit one copy of the gene from their parent without HD, and an expanded copy from their parent with HD. This means that for a person with HD, half their HTT protein is completely regular and unexpanded, while the other half is expanded.
The expanded HTT protein is thought to be the cause of HD-related symptoms. However, there are questions about what problems are caused by the presence of expanded protein, versus what problems are caused by the absence of enough unexpanded protein.

To address questions about the balance of expanded and unexpanded HTT protein levels in HD, researchers can use a variety of genetic tricks in mice, to manipulate where, when, and how much HTT protein is made, or “expressed,” in the brain and body.
Previous research in the HD field has shown that just expressing the expanded copy of the HTT protein for short periods during development in mice is enough to cause symptoms associated with HD when the mouse is older. But what’s also interesting is that the same thing happens when the normal, unexpanded copy of HTT is removed from the mouse during development!
Dr. Humbert’s team recently set out to explore in more depth the effects of losing or gaining different forms of HTT using specialized mouse models of HD.
Communication breakdown
Brain cells communicate, in part, by sending electrical signals throughout the brain. To study brain diseases, researchers will often measure these electrical signals to see how well brain cells are talking to one another.
Dr Humbert’s group ran experiments to compare electrical signals in the developing brains of mice with or without expanded HTT. The researchers found differences in these electrical signals when mice were very young. However, the electrical signals leveled out when the mice were older and eventually matched the mice with unexpanded HTT.
This seems to suggest the brain is able to compensate for changes in brain cell communication that it experiences early on in HD. The big question is – does this compensation last long enough and do these changes contribute to HD-related symptoms experienced later in life?
Cause and effect conundrum
HD researchers have long explored this cause-and-effect conundrum, questioning whether certain symptoms associated with HD are caused by the presence of the expanded HTT protein or loss of the regular HTT copy.
Dr. Humbert’s group asked if gaining expanded HTT or losing unexpanded HTT causes the changes they see in brain cell communication in their HD mice. They used creative molecular tools to turn off expression of only the regular HTT copy in nerve cells in the brain.
“Dr. Humbert’s team did some really cool science in mice to look at how both the expanded and unexpanded copies of huntingtin (HTT) affect mouse “symptoms” of Huntington’s disease (HD).”
Interestingly, nerve cells with lower amounts of the regular HTT protein responded in a similar way as those that express the expanded HTT protein, except their electrical signals didn’t level out over time. So it seems the brain cells without regular HTT did worse than those that have expanded HTT!
The researchers think this means that the HTT protein is necessary for communication between brain cells when mice are very young, and the presence of either form (expanded or regular) helps compensate as mice get older.
HTT affects nerve cell size and shape
Next the researchers looked at how the different forms of the HTT protein influence the shape of the nerve cells. Nerve cells are shaped like trees – with a cell body that contains many branches at the top, a long trunk, and a branched “root” system at the bottom of the cell.
They found that nerve cells from mice expressing the expanded HTT copy were less complex and had fewer branches when the mice were very young. As the mice got older though, they caught up in size and shape to match nerve cells from mice without the expanded HTT copy.
Interestingly, when they repeated the experiment that decreased the amount of regular HTT in nerve cells in mice, they had similar findings as before – they matched what was observed in the HD mice, except there was no compensation as the mice got older. This once again suggests that loss of regular HTT causes larger negative effects than expression of expanded HTT!
For this experiment they also added a drug called CX516 that increases the ability of nerve cells to send electrical signals. Using cool science, they added this drug to mouse embryos without HTT in their nerve cells before they were born. Excitingly, this drug improved the shape and size of the nerve cells. This suggests loss of the HTT protein affects the way cells communicate through electrical signals, but when that is restored, the brain can compensate!
Balance is key
The next question the researchers asked was if improving electrical communication in HD mouse brains with CX516 would affect HD “symptoms” in mice. They looked at different tests that examine mouse behavior, like how well mice can cross an open gap or how they travel through mazes. They found that CX516 improved how the HD mice performed on these tasks.
Interestingly, on all of the tests that looked at the effects of CX516 in mice without the regular HTT protein, the mice did worse. So even though CX516 increases electrical communication between brain cells, it seems that in mice without HD this does harm. These results highlight how delicate the communication circuits between cells are and show that tipping the scales too far in the other direction can also be bad.
It’s worth noting that the researchers won’t explore CX516 as a potential medicine for HD. It was previously studied as a possible treatment for Alzheimer’s disease, but it didn’t work very well. It’s more likely that they’ll look at ways to target brain cell communication in the same way CX516 does to further understand how the brain can compensate for HD-related changes.
Compensating for communication deficits
There are a few caveats to this work. The first is that CX516 was given to the HD mice before they were born. This undoubtedly leads to questions about how early we have to treat to see some of these effects.
While it might seem like this work suggests that we need to treat people that have expanded HTT almost as soon as they are born, that might not be the case. The brain is really good at compensation! There are lots of redundant pathways that ensure if something goes wrong, other mechanisms make up for it.
This is why the experiments in this paper saw a leveling off of electrical signals and changes in size and shape of nerve cells as mice got older – the brain was compensating. So treating HD when people are adults might still allow the brain time to compensate for changes related to HTT expression when people are young.
The second caveat to this work is that mice aren’t people! Seeing effects in mice is no guarantee that we’ll see the same thing in humans.
What we learned
Rather than studying HTT lowering for therapeutic gain, this new work by Dr. Humbert’s group lowered different forms of HTT as a tool – it allowed them to gain a deeper understanding of the effects caused by different forms of the HTT protein in mice that model HD. Studies like these could be very informative for therapeutics aimed at lowering HTT, giving us a more complete picture of what’s happening in HD.
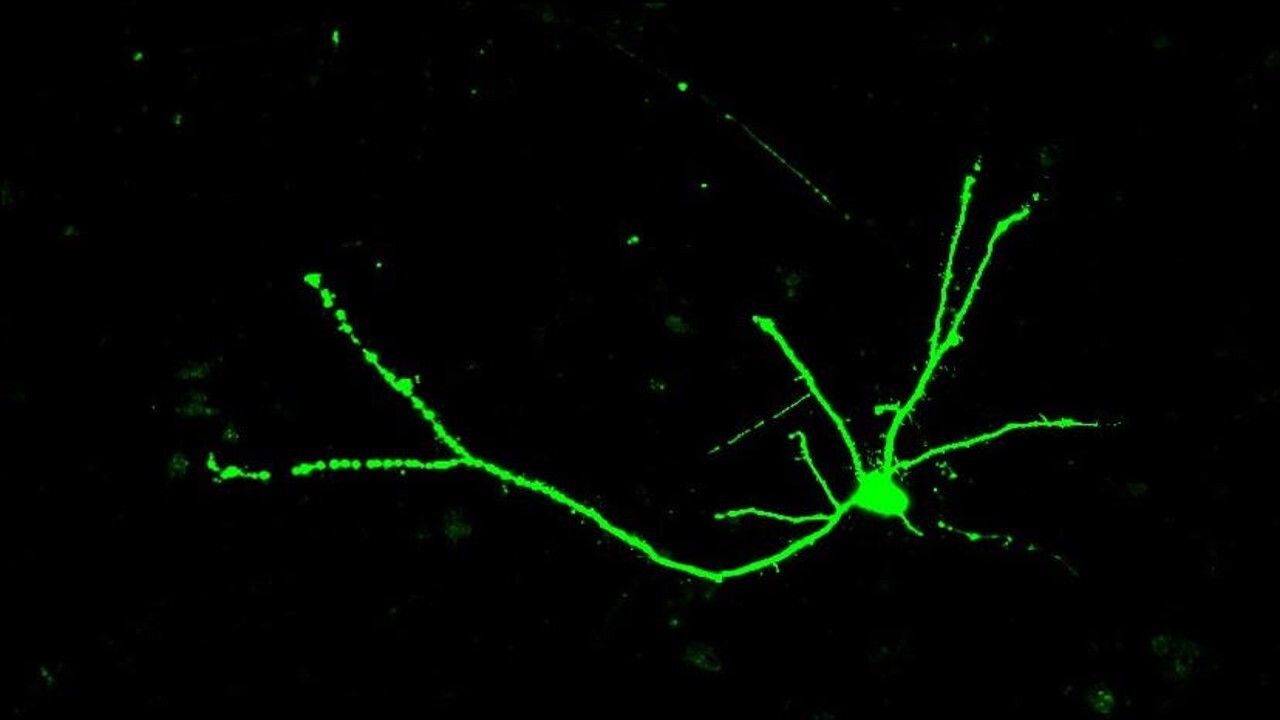
Image credit: Sarah Hernandez
This work also details some of the effects caused by the disease and the effects caused by the brain trying to compensate. Understanding that last bit – how the brain compensates – could help find balance in the brain and develop treatments for HD.
For more information about our disclosure policy see our FAQ…


