Stem cells and HD: past, present and future
Stem cells – who needs 'em? Scientists make brain cells from skin cells. Could this help HD?

Everyone’s heard of stem cells, but so far, stem cell treatments for Huntington’s disease have disappointed. Scientists can now create stem cells from skin samples – and even cut out the middle man to make brain cells directly. Stem cell treatments are still a long way off, but these cells are already accelerating HD research in the lab.
Irreplaceable neurons and atom bombs
Huntington’s disease is a neurodegenerative disease. That means that the symptoms are caused by the death of brain cells called neurons. Unfortunately for HD patients, neurons in the brain are generally not replaced once they die.
Image credit: Annie Cavanagh, Wellcome Images
How do we know this? For a long time, scientists had assumed it was true, based on studies in animals, but it was difficult to prove in humans. But in 2005 scientists did a remarkable experiment using radiation caused by atom bombs to show that neurons in the human brain were mostly not replaced.
From the mid-1940’s until a world-wide test ban treaty in 1963, hundreds of atom bombs were exploded in the earth’s atmosphere. These bombs released large amounts of a special type of carbon which can be distinguished from naturally occurring carbon. Measuring the amount of this carbon in neurons, and comparing it with the amount of carbon found in trees of a known age, scientists could assign the neurons a ‘birthday’.
The scientists determined that neurons in the brain had birthdays very close to the actual birthday of the person they belonged to. So, by and large, the neurons that you have when you die are the same ones you had when you were born. That’s one of the reasons why neurodegenerative diseases, like HD, are so damaging – cells that die are not replaced.
Stem cells and the dream of replacement
The fact that neurons are so important, and so irreplaceable, explains why many people are excited about stem cells. Stem cells are special cells that have the capability of turning into all the different cells that make up a body, from skin cells to liver cells to brain cells.
Every person alive started as a single cell – a fertilized egg. The cell divided, and the new cells went on to become all the many types of cells in the body. The ability to divide into cells of different types is called pluripotency, and was for a long time thought to be a unique feature of stem cells.
Stem cells are very difficult to come by. Historically, the only place that scientists knew to find them was the original source of all cells – the early stage embryo. A small mass of cells in the developing embryo could be isolated and grown in a laboratory. These cells are the ’embryonic stem cells’ that have caused such controversy and excitement all over the world. Embryonic stem cells can only be obtained from either terminated pregnancies, or the unused frozen embryos of couples going through fertility treatment.
The unique feature of stem cells to turn into other cell types made them very important to study, despite the difficulty in obtaining them. The dream of many people was that if we could understand how cells turn from one cell type into another, or ‘differentiate’, we could repair damaged tissues. If this were possible, we could replace dying pancreatic cells and cure diabetes, or repair breaks in the spinal cord and restore movement to paraplegics. Similarly, people hope that we could use neurons made from stem cells to replace dying neurons in the brain of patients with diseases like Huntington’s disease.
The challenge of replacing brain cells
The job of a neuron is to ‘talk’ to other neurons with electrical signals. That communication is key to all the remarkable things brains do. For example, the urge to move your finger starts as an electrical signal in a neuron in the brain near the top of your head. This reaches a neuron in your spinal cord, which then sends another signal all the way to the muscles of your finger. At the end of all this signaling, a puff of chemicals is released from the end of the neuron that started in your spinal cord, causing a muscle in your finger to contract.
You’ll have noticed that there are large distances being covered here. Only two neurons are directly involved in this movement from the brain to the tip of your finger. But cells are really small, aren’t they? So how do they reach such long distances?
Neurons grow extremely long extensions called axons that act like electrical wires to send and receive signals. These axons can be extremely long: giraffes have axons as long as 15 feet (4.5 metres)!
To fix damage in the brain, we need to think about these extensions, and all the different connections between neurons, as well as the brain cells themselves. Fixing the brain isn’t just about putting in more cells – the difficult part is hooking up the cells in the right patterns.
A simple finger movement involves a couple cells connected in a line. But some specialized neurons in the brain have hundreds of thousands of connections with other neurons. Getting the connections wrong could mean the entire network won’t compute correctly.
Stem cell trials in Huntington’s disease
In the hope that neurons might know how to grow new connections on their own, scientists have tried injecting immature human neurons, from embryo tissue, directly into the diseased areas of Huntington’s disease patients’ brains.
Results from these trials have been mixed. In one trial where five patients were treated, three of them found that their symptoms stabilized or even improved. However, the positive results didn’t last – the disease caught up and they started to deteriorate again.
“Fixing the brain isn’t just about putting in more cells – the difficult part is hooking up the cells in the right patterns.”
Why was the treatment disappointing? Probably for several reasons, the largest being that the cells just didn’t know what connections to make. But on top of that, the injected cells were not always perfectly healthy, and the HD brain they were being put into was also having problems already, which might have made it difficult for the cells to survive. In addition, the injected cells were genetically different from the brain they ended up in, which can cause the brain’s immune system to attack or ‘reject’ the cells.
At the moment, we just don’t know how to tell new neurons how to link up and make connections in adult brains. But because of these difficulties, replacing cells in the mature brain is unlikely to be a widespread therapy for HD any time soon.
So, are stem cells useless for HD?
These difficulties don’t mean that stem cells are irrelevant in HD.
To scientists, a problem like this is a challenge, and an opportunity to solve a mystery. We need to work with stem cells to try and understand the problem of making the right connections, with the long term goal of replacing cells in the brain.
Using animals, we can practice this and try to learn about the possibilities for re-growing connections between cells. Some day, this work might lead to technology that will enable the safe and accurate replacement of cells in the brain.
But even though we can’t use stem cells to treat Huntington’s disease yet, stem cells are important right now for helping us to understand and study HD.
The challenge of studying living cells
We have lots of good ideas for how the HD mutation damages neurons, but there are lots of things we still don’t know for sure. And understanding the problem is a key step on the road to solving it.
But it’s really difficult to do lab research on human neurons. Living human brain cells are very hard to get hold of – most alive people are still using their brains! And neurons don’t divide, so scientists can’t even take a few neurons and grow larger numbers.
Even if we had a source of adult human neurons, like tissue samples from brain surgery, neurons hate being removed from the brain and don’t don’t grow well in the lab.
We can grow neurons taken from young mice or rats, but even those are difficult to keep alive. Importantly, we know that there are huge differences between rodents and people, especially in the way their brain cells work.
Because of these difficulties, many of the cells used to study Huntington’s disease in the lab are tumor cells taken from various cancers. They grow well in the lab and are easy to handle. Using these cells moves research along more quickly, but of course HD isn’t cancer, and we might be misled by studying cells that are so different from the vulnerable cells in HD.
Stem cells as models of human disease
Stem cells can be encouraged to turn into any type of cell in the body, by treating them with different chemicals called ‘growth factors’. We’re getting better and better at understanding the procedures and recipes for making stem cells divide into various different cell types.
In fact, turning stem cells into neurons turns out to be one of the easiest things to do with them. Stem cells seem to ‘want’ to turn into neurons. Neurons made from stem cells can be used to try and understand what’s going wrong in Huntington’s disease, and try to fix it.
It’s in this area – basic lab work to study what goes wrong in cells with the HD mutation – that stem cells have the potential, right now, to revolutionize HD research.
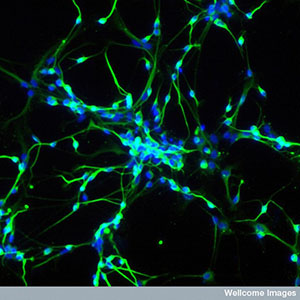
Image credit: Yirui Sun, Wellcome Images
Studying HD in the type of cells that die in the disease makes research findings much more believable – especially when the cells are human in origin. Recently, many major HD labs have begun working with neurons made from stem cells to better understand the disease.
And then, everything changed
Everything we know about stem cells changed in 2006. Two Japanese researchers, Kazutoshi Takahashi and Shinya Yamanaka, reported that they had been able to turn regular skin cells into stem cells. They figured out how to ‘re-program’ regular cells taken from a skin sample from an adult mouse, into cells that were indistinguishable from real embryonic stem cells. They called these new cells induced pluripotent stem cells or IPS cells.
With that single study, the idea that stem cells could only be obtained from embryos was basically changed over night. Suddenly, instead of future treatments using stem cells from embryos, we could imaging treating people with stem cells made from their own body – with their own DNA.
Of course, where stem cell treatment is concerned, the problem of getting the cell to make the right connections would still be there. But IPS cells could at least solve the problems of supply and the genetic difference between the injected cells and the brain.
Then it changed again
Just as scientists were getting used to the idea that stem cells might be much easier to obtain than they’d ever imagined, everything changed again. In 2010, a group of researchers at Stanford University did something maybe even more remarkable.
Rather than starting with adult cells, turning them into stem cells and then turning those stem cells into another cell type, they decided to cut out the middle man.
They showed that they could convert skin cells directly into neurons. Starting with a skin sample, they could grow cells in a dish, add a cocktail of chemicals and genes to re-program them, and the skin cells would turn into neurons – those irreplaceable brain cells whose loss underlies so many diseases, including HD.
Scientists have thought for decades that once a cell ‘commits’ to being one type or another, that decision can’t be changed. Clearly, that basic assumption is wrong.
Converting cells and HD
These remarkable advances – the ability to make pluripotent stem cells from adult patients, and the ability to directly re-program adult cells – have transformed the landscape of biology.
Things that seemed like science fiction five years ago are suddenly possible. Whatever the future holds, at a minimum, scientists can now study neurodegenerative diseases in human neurons, grown from real human patients.
But even with these new sources of neurons, we’re still left with the problem that simply injecting them into the brains of adults is unlikely to effectively replace neurons that die in disease. We need to find a way to re-grow the proper connections between them, that are critical for the proper functioning of the brain. That’s something Huntington’s disease researchers are working on, and it’s closer than ever – but still a long way off.
Meanwhile, the IPS cell revolution is just beginning to feed into our understanding of Huntington’s disease. As the techniques become more established, IPS cells will become crucial tools in the search for effective treatments.
Learn more
For more information about our disclosure policy see our FAQ…


