New tool to measure outcomes in Huntington’s Disease clinical trials
A better tool for HD trials! New research shows innovative way to measure mutant Huntingtin outside the brain
When patients participate in clinical trials, there needs to be some type of readout to determine whether the new treatment worked. It’s important to know two key things: What to measure and how to measure it. In the case of HD, these obstacles have vexed scientists and doctors for years. The latest research comes up with a clever new approach to overcome both challenges in a new way. These results could offer a valuable tool to study new HD therapeutics entering clinical trials.
What to measure and how to measure it: It seems straightforward. And sometimes, it is. For example, to test a new cholesterol-lowering medicine, doctors draw patients’ blood after treatment, measure cholesterol, and compare it to pre-treatment levels. In this case, the what is obvious (cholesterol), and the how is simple because cholesterol is a large, fat-like substance that can easily be separated from blood and measured.
But, in the case of HD clinical trials, what and how are much more complicated.
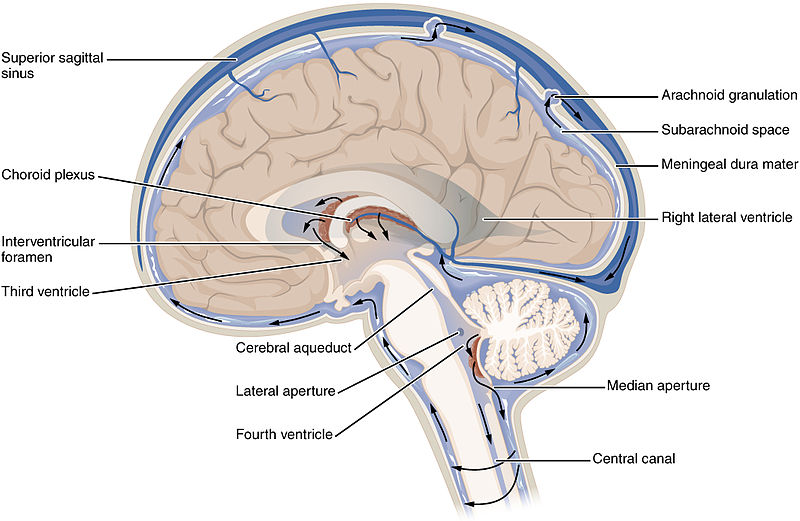
Image credit: Wikicommons
The “What”
HD is caused by a genetic mutation that changes the makeup of the Huntingtin protein, which makes the protein toxic. This mutant Huntingtin protein going rogue in neurons causes every case of HD. Mutant Huntingtin interferes with many vital neuronal functions, including transport of cellular cargo, gene regulation, and energy production.
Mutant Huntingtin normally lives inside of cells, including brain cells. But measuring mutant Huntingtin inside of the brain requires a brain biopsy—less than ideal, if you can avoid it. So, the hunt has been on to find less invasive ways of quantifying this tricky protein.
Remember I just told you that Huntingtin is normally found inside of brain cells? Well, that’s not always the case. Sometimes, mutant Huntingtin is released—possibly a result of neurons dying. When a brain cell dies, the membranes break down, allowing the contents to spill out. This means a lot of mutant Huntingtin that was once contained in the cell is now free. The brain quickly cleans up the mess left by the dying cell—but before being broken down, some of the contents travel through the fluid that bathes the brain, called cerebrospinal fluid.
The cerebrospinal fluid circulates throughout the brain and spinal cord. Doctors can insert a thin needle into the space below the spinal cord and withdraw small amounts of cerebral spinal fluid. By analyzing the fluid, doctors catch a small glimpse of what’s happening inside the brain without a brain biopsy.
“When the cells began to die, they observed a sharp spike in mutant Huntingtin levels in the cerebral spinal fluid.”
A new study from Drs Amber Southwell, Michael Hayden and colleagues asks, “Could we measure mutant Huntingtin levels in cerebral spinal fluid and use them to tell us about levels of mutant Huntingtin in the brain?” And the answer is yes!
Researchers have three pieces of evidence supporting this exciting new discovery.
They began by measuring mutant Huntingtin protein in the cerebral spinal fluid of HD mutation carriers. They drew samples from mutation carriers at various stages of the disease: advanced, early-mid, and before symptoms appeared. The researchers wanted to see if levels of mutant Huntingtin increase in cerebral spinal fluid as the disease progressed.
As they predicted, cerebral spinal fluid levels of mutant Huntingtin were highest in the people who were in more advanced stages of the disease, and lower in early disease stages. Why this is so is not clear – it may be that because more brain cells are dying in the course of HD, more mutant Huntingtin protein is released, but more studies will be needed to see if this is the case.
While that correlation was promising, researchers wanted to take things a step further to confirm that dying brain cells were really the source of cerebral spinal fluid mutant Huntingtin. So, they turned to mice.
Researchers poisoned a small area of brain cells. When the cells began to die, they observed a sharp spike in mutant Huntingtin levels in the cerebral spinal fluid. This could be because of the dying cells spilling their contents into the brain, and some of those contents, including mutant Huntingtin, traveling into the CSF.
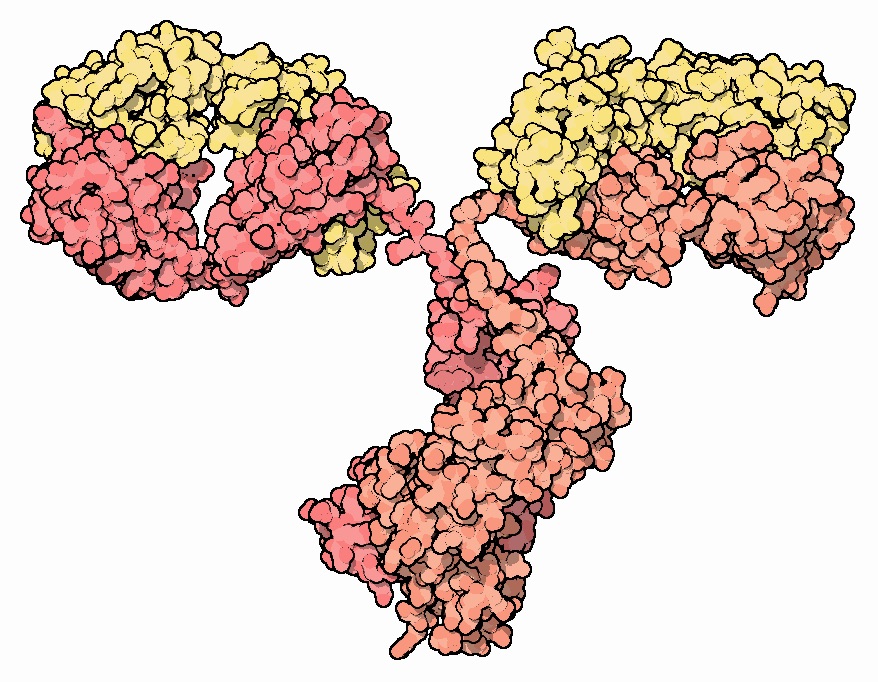
Image credit: Protein Data Bank
Huntingtin Silencing Tools
One of the most useful applications of measuring cerebral spinal fluid mutant Huntingtin would be testing new HD therapeutics. A promising avenue of research is to directly reduce the levels of mutant Huntingtin protein. For example, a new type of HD therapeutic approach called gene silencing aims to slow or stop disease progression by reducing levels of mutant Huntingtin. (Click here for an HDBuzz article about gene silencing: http://en.hdbuzz.net/023).
As a proof of concept, researchers treated HD model mice with gene silencing drugs that are known to reduce mutant Huntingtin levels in the brain. When they drew samples from the gene-silenced animals, they found levels of mutant Huntingtin in the cerebal spinal fluid had also been reduced! This suggests that measuring mutant Huntingtin in cerebral spinal fluid may be a good readout to measure whether gene silencing drugs in HD clinical trials are working.
The “How”
The idea of measuring mutant Huntingtin in cerebral spinal fluid has been around for a while, but it poses a huge technical challenge. As explained in this HDBuzz article (http://en.hdbuzz.net/197), mutant Huntingtin is just one of many proteins in cerebral spinal fluid. Isolating and measuring it is like finding a needle in a haystack.
“researchers planning human gene silencing trials have a powerful new tool to see how well these drugs are working”
But the other important discovery from this paper is a new, ultra-sensitive method of measuring mutant Huntingtin in cerebral spinal fluid.
The technique uses a pair of proteins called antibodies that specifically recognize and attach themselves to mutant Huntingtin and not other cerebral spinal fluid proteins. In fact, the antibodies are so specific that they only recognize toxic mutant Huntingtin, and not the normal, healthy Huntingtin, which is also produced in HD patients. Importantly, it can identify mutant Huntingtin floating through the cerebral spinal fluid on its own or bound to other proteins in multi-protein clumps, which is usually where it’s hanging out.
One of the antibodies in the pair is attached to an extremely tiny bead, and the other is attached to a glowing tag. The antibodies grab onto mutant Huntingtin. Then, the beads are collected, dragging mutant Huntingtin along with them. Finally, the beads plus mutant Huntingtin are fed through a special machine that can detect the light emitted from the tag, allowing the amount of mutant Huntingtin to be quantified. (More glowing = more antibody = more mutant Huntingtin.) This combination of highly specific antibodies and careful measurement of glowing light gave clinicians and researchers another reliable tool to measure mutant Huntingtin in cerebral spinal fluid.
This new technique complements another one recently described using similar methods, which found similar results (http://en.hdbuzz.net/197). Excitingly, this new study gives the first evidence that the mutant Huntingtin protein measured in the CSF originates in the brain, probably from dying cells. Most importantly, treatment of brains with drugs that reduce mHTT levels leads to quick changes in CSF levels.
This means that, whichever of these new techniques are used to measure HTT, researchers planning human gene silencing trials have a powerful new tool to see how well these drugs are working. We’re pretty excited about these trials, the first of which is just starting now, and this technique is a powerful new addition to testing these drugs in HD patients.
Learn more
Sources & References
For more information about our disclosure policy see our FAQ…