
Jumping genes: Huntington's disease protein invades brain transplants
A long-term study of the brains of HD patients who received grafts of fetal tissue reveals a surprising result

Huntington’s disease is caused by the malfunctioning and early death of brain cells. Replacing those dead and dying cells with stem cells has long been a goal of some HD scientists. A new study investigates the long-term health of some of the earliest cell transplants into HD patient brains — and finds a surprising result.
Filling in the gaps in the HD brain
Huntington’s disease, and other ‘neurodegenerative’ diseases like it, happen when specific cells in the brain die. Unfortunately for people carrying the HD mutation, these critical brain cells are mostly only made during our early development. After we’re born, most areas of the brain don’t grow many new brain cells to replace those that are inevitably lost, even during normal aging.
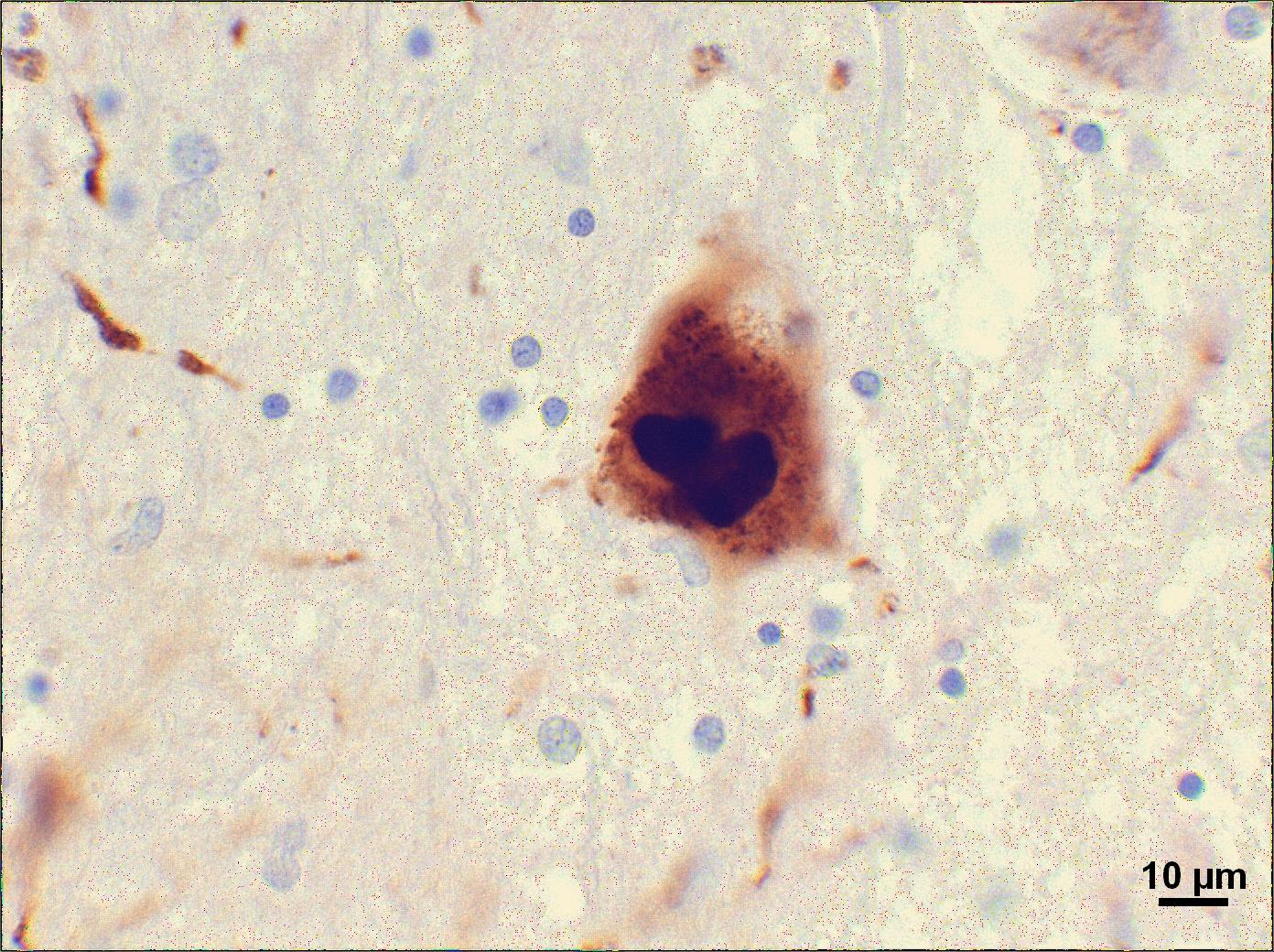
Image credit: Suraj Rajan
What if we could take tissue from a developing brain and use it to fill in the gaps in a degenerating HD brain? Though there’s a pretty high ‘ick-factor’, it’s technically possible to dissect brain regions from human embryos and transplant them into the degenerating regions of HD patient brains.
Cell replacement therapy
In fact, this ‘cell replacement’ idea has a long history in HD. In the mid-1980’s, a series of animals studies showed that it was possible to fix brain damage caused by toxins, by transplanting embryonic brain cells into the damaged area. Subsequent work, in more sophisticated animal models, supported the idea that this approach might be beneficial.
Based on this animal work, and the progression of similar trials in Parkinson’s disease, a small number of HD patients received grafts of embryonic brain tissue starting more than 15 years ago. Disappointingly, none of the patients receiving transplants showed much, if any, sustained improvement in their HD symptoms after receiving these transplants.
One patient who received a fetal tissue transplant died about 18 months after surgery, due to unrelated causes (heart disease). While sad for the patient and their family, this enabled scientists to study the transplanted tissue and see how it was doing in the brain. One possible explanation for why the patients didn’t get much better is that the transplants may not have survived, or might not have made the right kind of connections in the host brain.
In fact, this early study showed that the fetal tissue did survive in the brain of the HD patient, and the cells in the graft seemed to make the kinds of connections they should have made with other cells in the brain. This is good news, because it means this kind of transplant is technically possible, but bad news, because it means we don’t know why it didn’t make the patient better.
New cells, old problems
After more time had passed, scientists were able to study a larger number of brains from Huntington’s disease patients who had ultimately died of HD, years after receiving grafts of fetal tissue. This analysis pointed to a more disappointing reason for the failure of the grafted tissue to help HD patients: the new cells seemed to be dying, much like the old cells around them.
This was unexpected! Remember, the cells grafted into the brains of HD patients were from human embryos, and so were very young. Nevertheless, something about being inside of an HD brain made these brand new cells sick, and in fact lead them to die like the cells they’re supposed to be replacing.
Similar disappointing results were observed in Parkinson’s disease patients who’d received fetal tissue grafts, suggesting that this might be a general problem with the whole idea of cell replacement therapy. It could be that the brains of patients with neurodegeneration are just too inhospitable for new cells to be of much help.
There goes the neighborhood
But how could this be? If the donor cells don’t have an HD mutation, why do they get sick just like those cells that do? We don’t know the answer to this question yet, but an emerging body of work suggests brain cells in people with neurodegeneration may actually make each other sick.
“Cicchetti’s team noticed something strange about the grafted fetal tissue in HD patient brains — it contained aggregates! That’s very surprising, because this grafted tissue doesn’t have a mutant HD gene.”
In many neurodegenerative diseases, brain cells are found to be full of clumped up garbage. These clumps are called ‘aggregates’ in HD, ‘Lewy bodies’ in Parkinson’s and ‘amyloid plaques” in Alzheimer’s disease. In each case, cells in certain areas of the brain seem unable to take out the cellular trash, which might contribute to them getting sick and dying.
When fetal grafts were implanted into the brains of patients with Parkinson’s disease, the cells in the graft were discovered to contain Lewy bodies, just like the sick cells around them. This was very surprising – these are healthy young cells, and it normally takes decades for Parkinson’s disease to develop.
New HD work
Could something similar be happening in Huntington’s disease grafts? A recent study from a group of scientists led by Francesca Cicchetti, Université Laval, suggests something funny might be going on. Cicchetti examined the brains of 3 HD patients who died about 10 years after receiving grafts of fetal tissue.
To understand their findings, we have to remember a few things about how HD works. Every HD patient has inherited a mutant copy of the HD gene, which causes their cells to make a mutant HD protein. It’s this mutant HD protein that causes damage in the HD brain. In fact, most of those clumps of garbage found in HD brain cells (the ‘aggregates’) are made of the mutant HD protein.
Cicchetti’s team noticed something strange about the grafted fetal tissue in HD patient brains ― it contained aggregates! That’s very surprising, because this grafted tissue doesn’t have a mutant HD gene, and so shouldn’t have any mutant HD protein in it. What’s going on?
To be clear: the clumps of mutant HD protein aren’t inside the cells of the graft, but rather stuck outside the cells like litter that shouldn’t be there. The explanation for this surprising result isn’t clear, but figuring out where these clumps come from and whether they contribute to the failure of these grafts is going to be an important area of work. But at least now we know that they’re there.
So, now what?
The results of this study, as well as the other studies in other neurodegenerative diseases, suggest that we need to be very cautious about simply replacing dead cells in the degenerating brain. If the underlying sickness is still present, the new cells we put in the brain may simply become ill as well.
This is somewhat disappointing news, in terms of accomplishing cell replacement therapy for HD. But great strides are being made in stem cell science in labs around the world, so this story is not the end of this particularly road. Finally, though cell replacement is an attractive idea, work to boost the survival of brain cells, rather than replacing them when they die, is rapidly developing and continues full speed.
Learn more
For more information about our disclosure policy see our FAQ…


