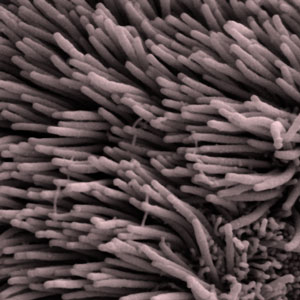
HD just got cilia
Huntington's disease just got cilia: huntingtin protein affects tiny but important hairs on cells called cilia

A better understanding of the normal role of the huntingtin protein would make developing treatments easier. Surprising new results from French researchers suggest that huntingtin influences tiny hair-like structures called ‘cilia’. Now we need to figure out what it means for patients.
Who is Cilia?
Look at a drop of pond water under a microscope and you’ll see thousands of tiny single-celled animals swimming around. These organisms need to swim towards food and away from predators. They’re coated in rhythmically beating hairs called cilia that power their movement.

Some human cells have cilia, too. For example, the air passages of our lungs are lined with cells that have cilia. The constant waving of the cilia pushes dust and unwanted gunk out of the lungs.
As well as these actively waving cilia, virtually every cell on the body has a single, non-moving hair. These are called the ‘primary cilia’. We don’t fully understand what primary cilia do, but they may act a bit like an antenna for cells, perhaps helping pass information from outside the cell to the inside.
Did Huntington’s disease just get cilia?
This would seem like a pretty obscure bit of biology, except for the fact that problems with the primary cilia have been linked to human diseases. In recent years, several diseases that were previously unconnected have been linked because the mutant genes that cause them are part of the cilia complex.
We know that the Huntington’s disease genetic mutation makes the huntingtin protein become harmful, and that’s what ultimately causes disease symptoms. But the non-mutant huntingtin protein keeps surprising us, too.
We don’t fully understand exactly what huntingtin does, but we know it’s important. Mice genetically engineered to have no huntingtin at all don’t survive long enough to be born.
Having a better understanding of the normal function of the huntingtin protein might help us to understand the disease process.
Frédéric Saudou of the Institut Curie in Paris has a long standing interest in the normal function of the huntingtin protein. While looking at where the huntingtin protein ended up in cells, they noticed that it was often found in the same place as the primary cilia. In light of the recent interest in cilia in disease, they decided this was worth following up with more studies.
Lack of huntingtin causes problems with cilia
Saudou’s team used a technique called RNA interference or RNAi to reduce the levels of huntingtin in cells from mice. RNAi is a form of gene silencing that lets researchers ‘turn off’ individual genes.
“In mice whose ependymal cells don’t have any huntingtin, there are serious problems in the brain”
They figured that if finding huntingtin in the cilia was just a coincidence, the cilia wouldn’t mind if the huntingtin was taken away. Instead, they found that when they reduced the huntingtin levels, the number of cells with cilia fell dramatically.
This is cool evidence that huntingtin, as part of its normal role in cells, helps the cilia to form.
But what happens in living mice when huntingtin levels are reduced? To study this, Saudou’s team engineered a mouse that didn’t produce any huntingtin in a single type of cells – ependymal cells. When you’re showing off to your friends, make sure you emphasize the second syllable – epENdymal cells.
Our brains contain fluid-filled spaces called ventricles. They’re lined with sheets of ependymal cells, and it’s the ependymal cells that secrete the fluid that circulates around the brain carrying messages and nutrients. That fluid is called cerebrospinal fluid.
Ependymal cells are of particular interest here, because they use cilia on their surface to help the cerebrospinal fluid to circulate.
When Saudou’s team studied the mice whose ependymal cells didn’t have any huntingtin, they found serious problems in the brain. As predicted from the work in cells, the cilia of the ependymal cells didn’t form properly without huntingtin. More importantly, the mice developed a serious problem of the brain called ‘hydrocephalus’ – high pressure in the head due to a buildup of fluid.
Clearly, having huntingtin help cilia do their job is important for brain development.
Mutating huntingtin causes cilia problems too
This is interesting, but HD patients don’t have a lack of huntingtin – they have mutated huntingtin. So what happens with cilia when huntingtin is mutated?
To investigate this, Saudou turned to cells and mice that had been genetically modified to have a mutant HD gene. In cells from these mice, the cilia were longer than normal, and there were more of them. That’s the opposite of what had happened when huntingtin was reduced.
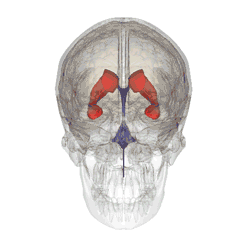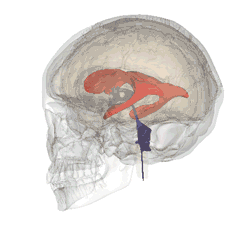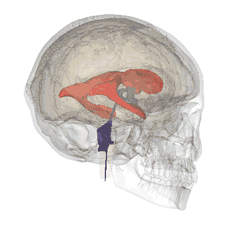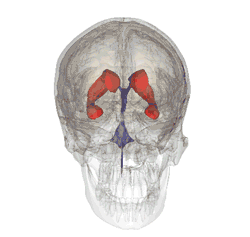
Image credit: Life Science Databases
Back to patients
When things get this weird in biology, it’s always a good idea to go back to patients and see whether your unexpected results are relevant. So Saudou’s team turned to brain samples donated by HD families. When they looked at the cilia of ependymal cells in HD patient brains, they discovered that they were longer, exactly as they’d seen in the mouse cells with the mutant HD gene.
If the job of ependymal cells is to keep the fluid in the brain moving around, what happens when the cilia on these cells are longer than they’re supposed to be?
In an attempt to answer that question, Saudou’s team returned to the mutant HD mice. They added small visible particles to the cerebrospinal fluid and watched them move around. The flow of the fluid around the brains of the HD mice was severely disrupted, and slower than normal.
Interesting, or important?
Anything we can learn about Huntington’s disease helps bring us closer to the day when we can treat it. While this work is extremely well done, it might not be obvious why it’s relevant to HD families.
Any time we can link together different diseases – for example, linking Huntington’s with other diseases where there are problems with the cilia – we can combine resources with other fields. Many tools and drugs have been developed in these other fields that might help people studying and living with HD.
Fundamentally new insights into how the huntingtin protein works don’t happen very often, and are worth celebrating when they do.
Learn more
For more information about our disclosure policy see our FAQ…


