Latest News
-

HDSA FAQ on the Roche/Genentech RG6042 program
HDSA's frequently asked questions factsheet about the huntingtin-lowering trial ASO program was so good we stole it (with permission)
-

Kids sometimes get Huntington’s disease too
Finally, a big study that shows what childhood HD looks like. This will help us work out if new drugs work in children too
-

Roche announces details of its 'pivotal' huntingtin-lowering study
The GENERATION-HD1 trial will test whether RG6042 – formerly Ionis-HTTRx – slows the progression of Huntington's disease
-

Disappointing news from LEGATO-HD trial of laquinimod in Huntington's disease
The LEGATO-HD trial of laquinimod did not slow progression of Huntington's disease. Here's the lowdown.
-

HDBuzz Enrolls in Enroll-HD
As promised, HDBuzz's Jeff Carroll finally signed up for the Enroll-HD study. Learn more about this important global HD "observational study" here.
-

First Dolly, now Piglet; a new Huntington’s disease pig knock-in model confirmed
For the first time in HD research, an animal model has been created in pigs using 2 cutting edge DNA editing t
-

New collaboration seeks to speed Huntington's disease drug licensing
Critical Path Institute launches new initiative to get HD drugs licensed as quickly as possible
-
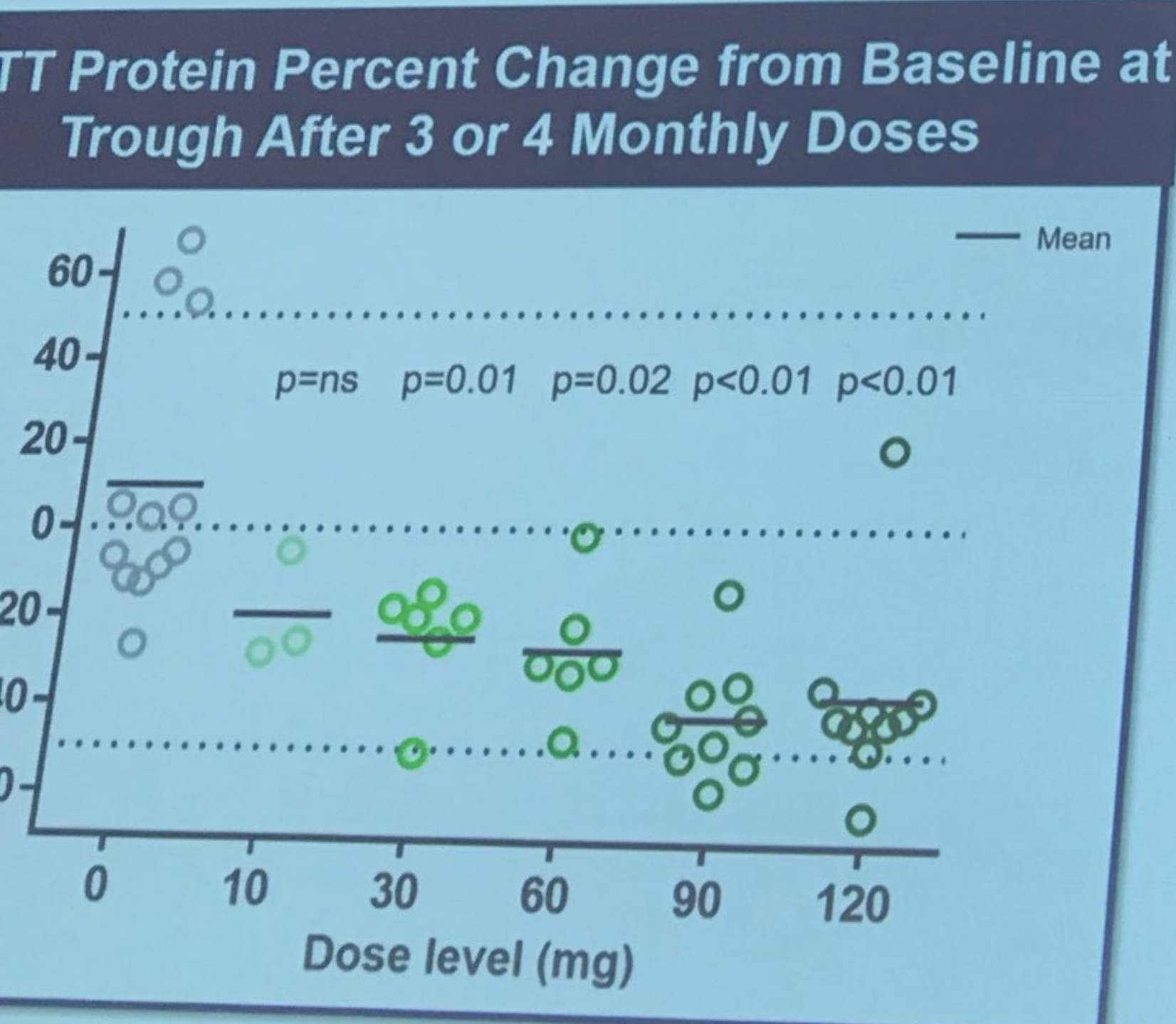
HDSA’s Q&A on the latest huntingtin-lowering update from Ionis and Roche
Our friends at @hdsa have produced this really good Q&A document about the latest huntingtin-lowering news.
-

Exciting Experiments in "Open Science" by Huntington's Researchers
Brave scientists open their notebooks to help drive HD research at the risk of getting "scooped".
-

The structure of the protein that causes Huntington's disease, revealed
Huntingtin protein's structure is now clear thanks to cryo-electron microscopy. This is kind of a big deal








