
Researchers with PTC Therapeutics recently published exciting new findings - a promising new huntingtin lowering drug that can be taken as a pill. Will this change how we move forward with huntingtin lowering?

Researchers show that highly expanded CAGs in the HD gene can cause early developmental changes using 3D brain models called organoids. What’d they find?
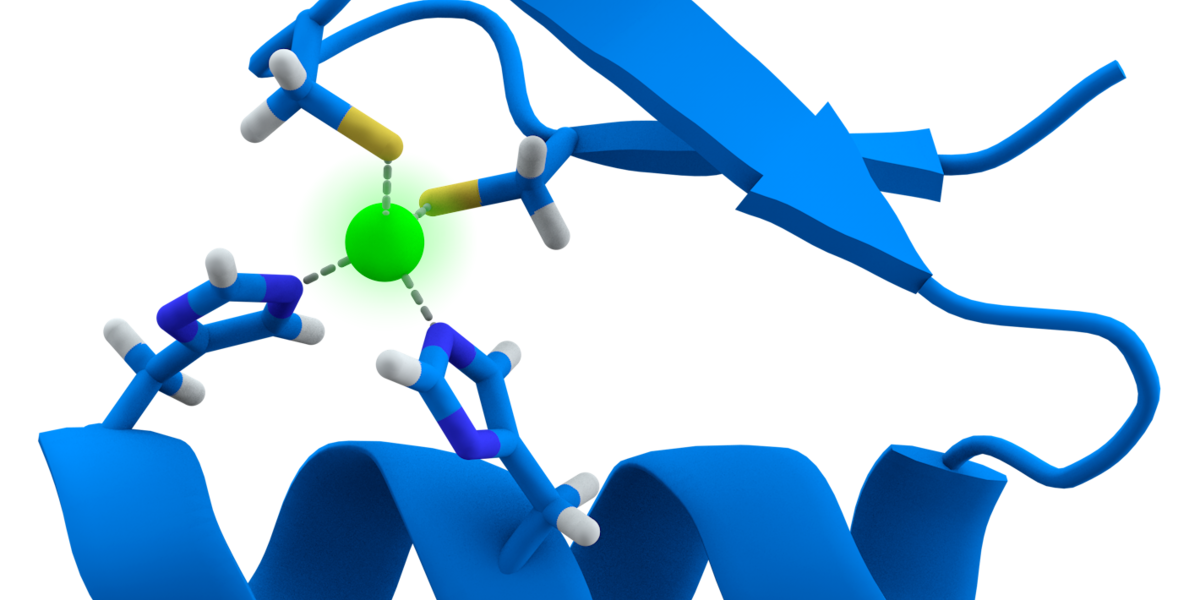
Exciting new Huntingtin Lowering work from @SangamoTx and @CHDIfoundation using "Zinc Fingers" to shut down expression of the mutant Huntingtin gene. More details on this exciting new technique here.
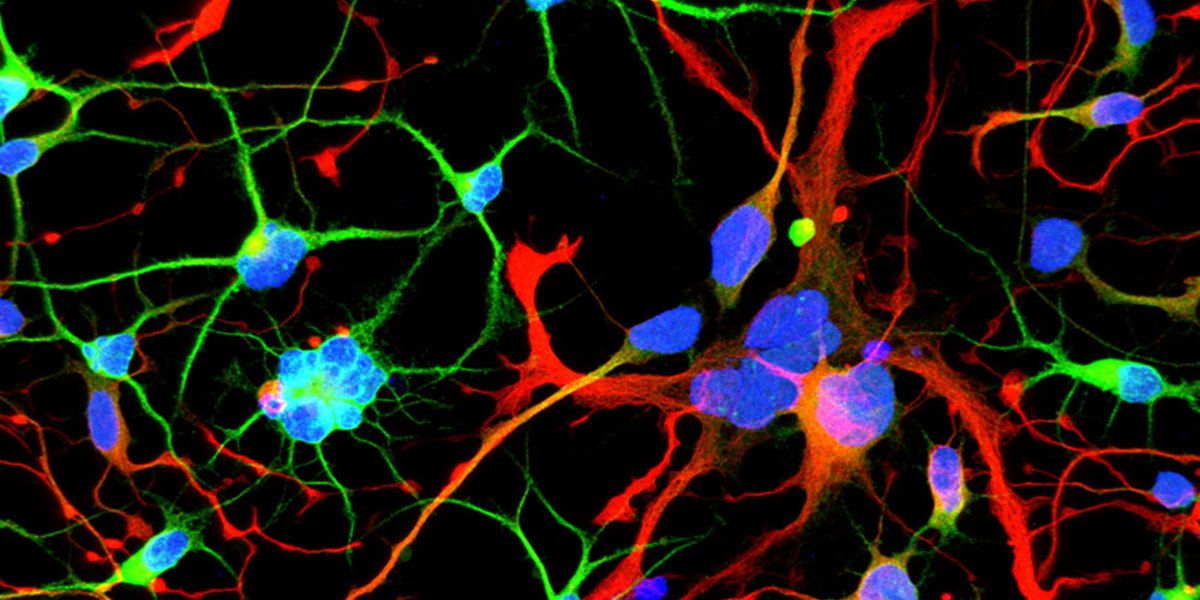
A surprising new paper sheds light on the role of the HD gene early in development. Should we worry?

Media reports of a wonder drug for neurodegenerative diseases like Huntington's disease are overhyped

Two recent studies show how a cellular border control system goes wrong in HD, opening new avenues for HD research.

Huntingtin helps fix damaged DNA, a recent study suggests, under the direction of a gene repair protein called ATM.
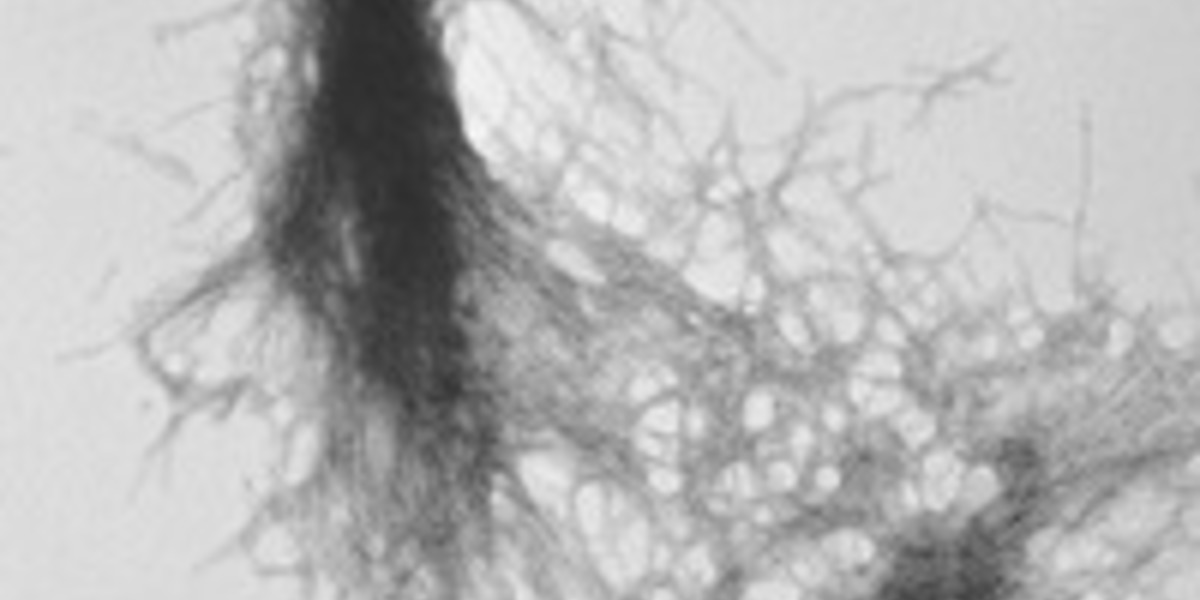
Harmful misfolded huntingtin can travel between brain cells via messenger particles called exosomes

HD brain fluid makes brain cells grow clumps.